Research identifies process to make cellulose in tightly bonded lignocellulosic bagasse and cachaza substrates available to microorganisms for anaerobic degradation, without expensive pretreatment.
Avijit Dasgupta
BioCycle September/October 2019
Processing sugarcane for manufacture of sugar generates two significant lignocellulosic solid wastes — bagasse and cachaza. Bagasse is the fibrous residue of the sugarcane stalks after juice extraction, and cachaza is the lime-filter mud from the sugar refining process. Every 1,000 tons of sugarcane processed produces approximately 100 tons of sugar, and generates approximately 250 to 270 tons of bagasse, and 30 to 35 tons of cachaza.
Part of the bagasse generated is often burned as fuel in the sugar mill boilers, which results in air pollution — including particulates, greenhouse and other gas emissions — and requires installation of expensive stack scrubbers to clean the emissions to comply with air quality regulations. In some cases, a portion of the bagasse may be used as raw material for manufacture of paper. Most of the bagasse and cachaza are disposed offsite in open waste piles causing rodent infestation, groundwater contamination from leachates, and land and surface water pollution from runoff. Degradation of the bagasse waste in waste piles results in greenhouse gas emissions, also causing air pollution.
In a highly competitive world market, disposal of large quantities of sugarcane process solid wastes (bagasse and cachaza) is also a cost burden for the sugar industries, as is the high cost of energy in manufacturing operations. Sustainability of sugar industries, particularly for the small and medium sized facilities, is a major challenge, while thousands of tons of bagasse-cachaza wastes, a potential resource for beneficial use, remain unused and pollute our environment.
Research Objectives
The objective of the investigation was to evaluate an alternative approach to utilize the bagasse and cachaza lignocellulosic wastes to address the environmental and economic issues faced by the sugar industries. The alternative solution was to anaerobically process a mixture of cellulose-rich bagasse and the cachaza wastes as a substrate to generate methane-rich biogas to be used in the sugar mill boilers. It offers the advantage of using the biogas as a clean fuel to provide a source of heat critically required in sugar manufacturing and eliminates or significantly reduces the use of fossil fuel. One of the key challenges was to find a relatively cost-effective way to make the cellulose in the tightly bonded lignocellulosic substrate available to the microorganisms for anaerobic degradation, without expensive pretreatment.
Materials And Methods
Developing suitable laboratory-scale anaerobic digestion (AD) units was an important part of the initial set up for the laboratory studies. Six cylindrical plexiglass digesters, each, 4.5 liter capacity, were used. Baffle plates were installed in the digesters to ensure dispersed homogeneous mixing conditions of the contents. Double-lid digester covers fitted with an O-ring were used to prevent leakage of gas from the digesters. Gas collection tubes fitted to the lids were connected to an inverted graduated cylinder filled with water and immersed in a water bath. The volume of gas produced was measured by the displacement of water in the graduated cylinder. The digesters were partially immersed in a water bath of constant temperature (35°C). Magnetic stirrers were used for continuous mixing of the digester contents. Attaching baffle plates to the digester walls ensured satisfactory mixing and prevented any vortex effects.
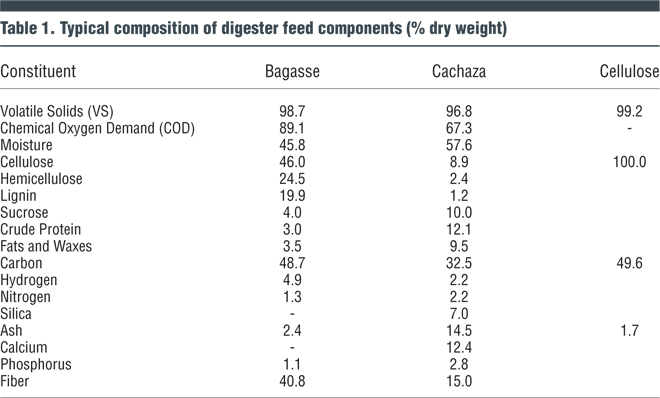
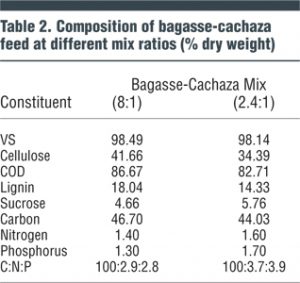
The substrate used for the experiments was a mixture of bagasse and cachaza obtained from a sugar mill. The bagasse was dried and milled to a size of Mesh 20 sieve. Two mix ratios of bagasse and cachaza were investigated. One was to represent the quantities of bagasse and cachaza typically generated daily (8:1 dry weight) in sugar mills; the second mix (2.4:1) represented the relative proportion of bagasse and cachaza after 70 percent of the bagasse produced was incinerated as a boiler fuel in the sugar mill. Composition of the substrate mixes is given in Tables 1 and 2.
Although bagasse and cachaza contain high concentration of cellulosic organic matter (96% volatile solids), its availability for microbial degradation is limited mainly by two factors — the complex non-hydrolyzed form of cellulose, and the binding effect of lignin to the cellulose, which deters it from being readily available for hydrolysis. Considering the challenge of the lignocellulosic substrate for microbial degradation, acclimation procedures were conducted to prepare an active mix of microbial culture for conducting the investigations. Since a heterogeneous culture of microorganisms would be required, a mixture of freshly digested anaerobic sewage sludge, cow manure and soil containing decaying leaves from under trees was used to seed the bagasse and cachaza mixed substrate.
A homogenized slurry of filter paper was added to the seed-substrate mix daily for two weeks. Gas production was monitored. This procedure facilitated a smooth transition for the microorganisms to acclimate to the lignocellulosic substrate. The slurry of seed and substrate mix was used as the acclimated culture for the two types — batch and semi-continuous — of laboratory AD investigations. For the initial batch studies, water was added to the substrate mix based on the organic loading investigated to prepare the feed slurry. For the semi-continuous reactor studies, investigations were conducted with water and using recirculated filtrate to evaluate the effect of recirculated filtrate on the digester performance. Several parameters (pH, alkalinity, volatile solids, COD, and nitrogen) were monitored. Gas composition was monitored in the gas generated for methane and carbon dioxide using a gas chromatograph with a thermal conductivity detector.
Initial Batch Digestion Studies: Before conducting semi-continuous digester investigations, batch studies were done. The objective was to determine the amenability of the bagasse-cachaza substrate to anaerobically degrade and produce methane gas. The investigations were conducted with bagasse and cachaza separately and as a mix. The results indicated that using the acclimated microbial culture, the bagasse and cachaza mixed substrate is amenable to anaerobic degradation and produces methane-rich gas.
Semi-Continuous Digestion Studies: Based on the promising results of the batch digestion studies, further investigations were conducted using semi-continuous anaerobic digesters. The objective was to develop a closed-loop system for efficient bioconversion of the bagasse-cachaza mixture to generate methane-rich biogas. These investigations were carried out on a battery of six digesters operated as completely mixed semi-continuous reactors with daily withdrawal and loading of digester slurry. The digesters were operated at mesophilic temperature conditions (35°C), similar to the batch studies. Different types of investigations were conducted to evaluate the effects on the following conditions: Organic loading and recirculation of digester filtrate; Detention time; Substrate composition; and Gas composition.
Results
Organic Loading And Recirculation Of Filtrate
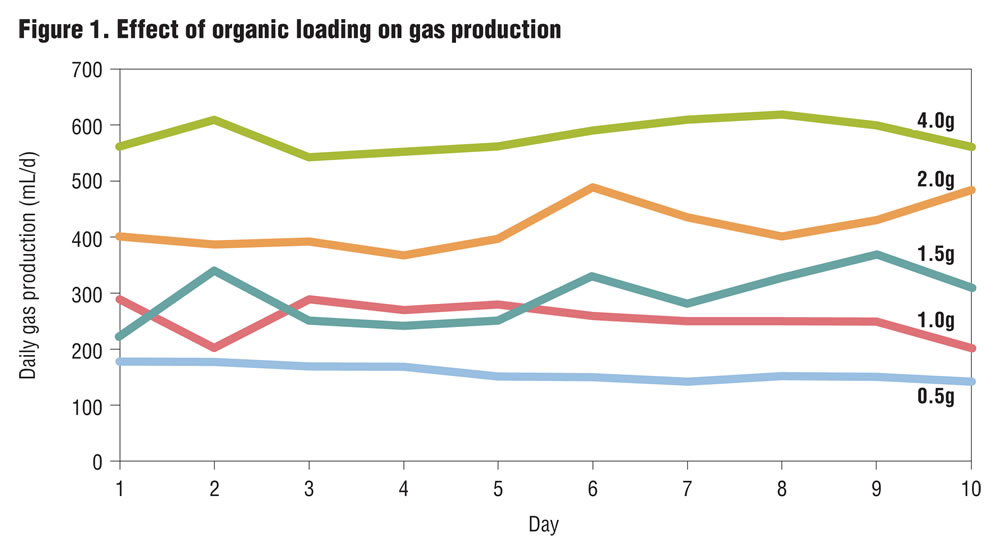
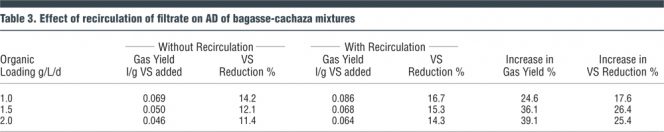
The semi-continuous fully mixed digesters were operated with a detention time of 10 days. The effect of organic loading rate on gas production was studied. Five different organic loading rates were used: 0.5, 1.0, 1.5, 2.0, and 4.0 gm volatile solids/liter/day (VS/L/d) using digesters operated in duplicate. The bagasse-cachaza mix used (8:1 by dry weight) for the investigations represented the operating condition in the sugar mill with no utilization of the bagasse as fuel in the boilers. Results of these studies are presented in Figure 1, which shows that the digesters with increased organic loading had higher gas production compared to ones with lower organic loading. The gas yield ranged from 0.036 L/g VS added to 0.079 L/g VS added. The volatile solids reduction ranged from 8.1 to 17 percent.
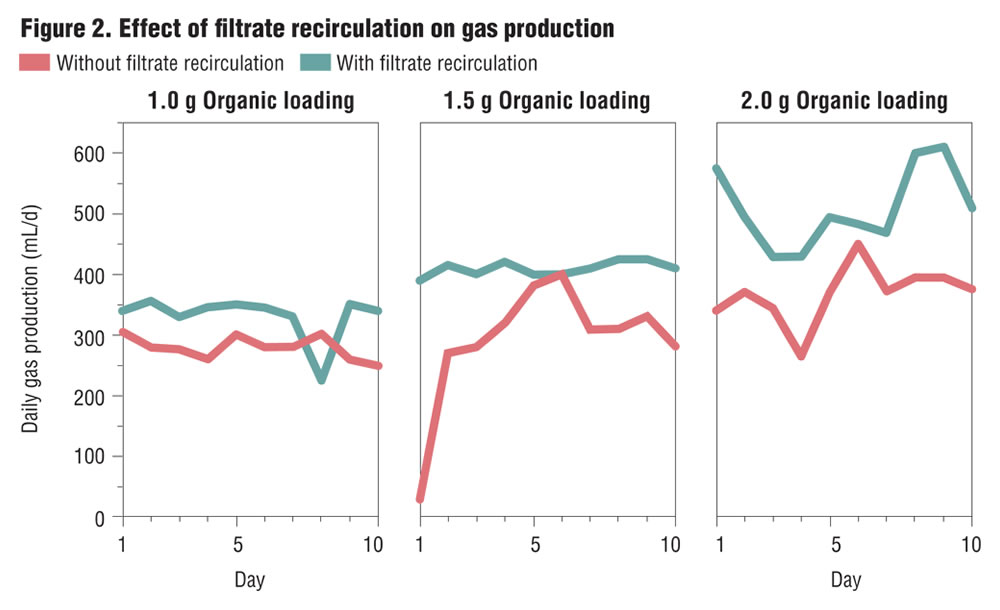
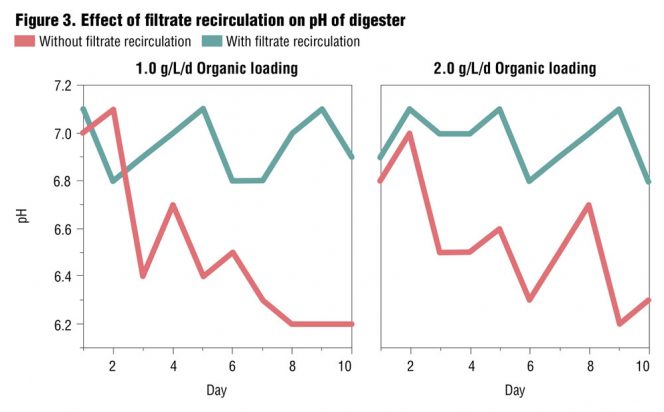
Based on these results it was decided to investigate if recirculation of the digester filtrate would improve digestion efficiency and facilitate increased gas production. The digester slurry was filtered, and the filtrate was recirculated in the subsequent investigations. Organic loadings of 1.5 and 2 g VS/L/d were selected for the recirculation investigations. The results of these investigations are shown in Figures 2 and 3, and indicate that recirculation of the filtrate had significant effects in improving digestion performance. Recirculation resulted in increased gas production (26-39%), a more stable operating pH condition in the digester, increased volatile solids reduction, and higher gas yield per unit of volatile solids reduction (Table 3). Based on the results, further investigations were conducted with filtrate recirculation.
Detention Time
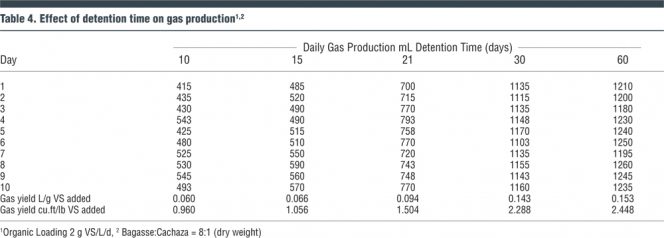
The purpose of this phase of the research was to determine the effect of detention time on gas yield for a constant organic loading. Bagasse-cachaza mix (8:1) at an organic loading of 2 g VS/L/d was used as substrate to conduct a series of digester runs with detention times of 10, 15, 21, 30 and 60 days. Digester filtrate was recirculated. Results of these investigations are shown in Table 4. The optimum detention time was determined to be 30 days (based on gas yield) beyond which there was no substantial improvement in gas production. Volatile solids reduction also increased considerably for a change of detention time from 10 to 30 days. The gas yield increased marginally for a 60-day detention time. Moreover, providing a 60-day detention time would require a very large size digester at high cost without major benefits.
Substrate Composition
As mentioned above, one of the common operating conditions of sugar mills evaluated was when 70 percent of the bagasse is burned as fuel in boilers and the remaining quantity (30%) of bagasse, along with the entire quantity of cachaza, is disposed offsite and available for AD. This condition represents a bagasse-cachaza substrate composition of approximately 2.4:1 (by dry weight). Further investigations were conducted using this 2.4:1 substrate composition. Based on detention time studies, investigations to compare the two substrate mixes (the 2.4:1 versus the 8:1 substrate mix) were conducted using a 30-day detention time. The effect of organic loading on gas production and other digester performance parameters was investigated for organic loadings of 0.5, 1.0, 1.5 and 2.0 g VS/L/d.
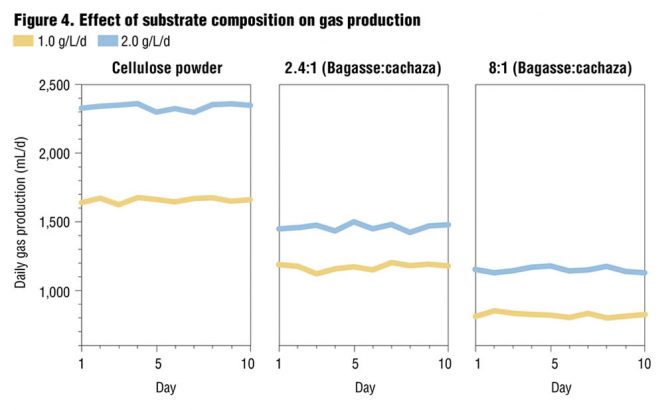
Digestion was conducted to compare the effect of lignin present in the lignocellulosic bagasse-cachaza substrate mixes on the AD process. The results, presented in Figure 4, indicate that for an organic loading of 1 g VS/L/d, a higher gas yield of 0.293 L/g VS (4.6 cu ft/lb VS) was obtained with the 2.4:1 substrate mix compared to that of the 8:1 substrate mix (generating gas of 0.205 L/g VS or 3.2 cu.ft./lb VS).
Studies were also conducted to compare gas yield of the bagasse-cachaza mix with pure cellulose substrate (Figure 4). In the case of cellulose, for similar organic loadings, much higher gas yield (0.414 L/g VS or 6.5 cu.ft./lb VS) was obtained than with the bagasse-cachaza substrate mix. The lower gas yield obtained in the case of bagasse-cachaza mix relative to the cellulose substrate is attributed to the presence of lignin in the bagasse-cachaza substrate. The lower volatile solids reduction for the bagasse-cachaza substrate compared to the cellulose also supports this conclusion. Volatile solids reduction was higher (37%) in the 2.4:1 mix than the 8:1 mix (29.1%). Cellulose substrate had the highest volatile solids reduction (47.4%). Based on these results, organic loading of approximately 1 to 1.5 g VS/L/d with a bagasse-cachaza mix of 2.4:1 and 30 days detention time, with filtrate recirculation, appears to be a suitable condition of substrate for anaerobic digestion of the bagasse-cachaza mix substrate investigated.
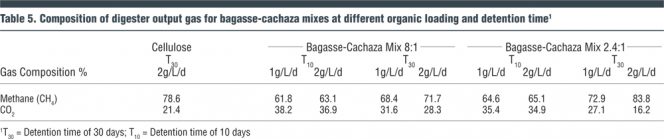
Gas Composition
The gas produced was analyzed for methane and carbon dioxide (CO2) during the different digestion runs (Table 5). The gas composition indicated methane and CO2 as the two main constituents. Hydrogen sulfide was not detected. The average concentration of methane for a 30-day detention time was approximately 78 percent for the 2.4:1 substrate mix and 70 percent for the 8:1 substrate. In general, methane content in the gas increased with longer detention time. The relatively high concentration of methane in the gas offers a good opportunity for it to be used as an efficient clean boiler fuel and heat generation thus saving fossil fuel use and reduced cost of energy for sugar processing in the mills. Absence of hydrogen sulfide is an additional advantage since the gas will not require cleaning for use as boiler fuel and will not produce toxic gas emissions causing air pollution.
Conclusions
The investigations conducted on the anaerobic digestion of a bagasse-cachaza mixture support the following conclusions:
-
- • It was possible to develop a microbial culture acclimated to the bagasse-cachaza substrate, effective for mesophilic AD of the substrate to produce methane-rich biogas without pretreatment.
- • The AD system developed based on this investigation is a closed-loop operation with zero waste. In addition to production of methane-rich gas for use in the sugar mill, elimination of fossil fuel use and abatement of pollution of land, water and air, the system reuses the digester filtrate to improve the stability and gas production of the digestion process, and uses the digester’s filter cake as a fertilizer supplement on the sugarcane farms, reducing fertilizer costs. Based on this investigation, biogas of approximately 4 to 5 cu.ft./lb. of dry volatile solids is produced. Utilizing the biogas as fuel, a 10,000 tons/day sugarcane processing mill can potentially generate approximately 9,000 million BTU of heat energy/day or 2 million kWh of electric power.
Existing pretreatment technologies and subsequent processes that are required prior to AD when using lignocellulosic agricultural wastes for bioenergy pose a significant cost burden that limits the economic viability of an AD system with pretreatment. The findings reported in this article offer a promising opportunity for the cane sugar industry to conduct pilot-scale investigations and implement full-scale AD systems without pretreatment to achieve cost savings and environmental benefits for sustainability of their operations.
Avijit Dasgupta, PhD, P.E., is a Senior Vice-President at Enviro Associates International in Commack, New York; dasgupta.enviro@yahoo.com
Bibliography
Chynoweth, D.P. and Ghosh, S., ‘Anaerobic Processes’. Annual Literature Review. Jour. Water Pollution Control Federation 51, 1200 (1979).
Keenan, John D, and Kormi, I., ‘Anaerobic Digestion of Brewery By-products’. Jour. Water Poll. Control Fed., 53, 66 (1981).
Ghosh, S and Klass, D.L., ‘Advanced Digestion Process from Biomass-Waste blends’. Symp. American Chem Society, 144, 251 (1981).
Cowling, E.B., ‘Physical and Chemical Constraints in the Hydrolysis of Cellulose and Lignocellulosic Materials’. Biotech. Bioeng. Symp. 5, 163 (1975).
Gaddy, J.L. et al., ‘Kinetic Improvements in the Production of Methane from Cellulosic Residues’. Proc. Symp. Energy from Biomass and Wastes, Florida (1983).
Bhadra, A., Scharer, J.M. & Moo-Young, ‘Anaerobic Digestion of Native Cellulosic Wastes’. M. Mircen Journal (1986).
Adney, W.S., Rivard, C.J., Shiang, M. et al. ‘Anaerobic Digestion of Lignocellulosic Biomass and Wastes’. Appl Biochem Biotechnol, 30:165 (1991).
Pfeffer, J.T. & Liebman, J.C., ‘Bioconversion of Municipal Solid Waste to Methane’. Ann. Rpt. to NSF Grant G139191 (1974).
Dasgupta, A, ‘Anaerobic Digestion of Municipal Solid Waste’. Biocycle 22, 2 (1981).
Slonekar, J.H., ‘ Agricultural Residues, Including Feedlot Wastes’. Biotech Bioeng, 6, 235 (1975).
Foutch, G.L. & Gaddy, J.L., ‘Culture Studies on the Conversion of Cornstover to Methane’. Bioeng Symp. 11, 249, (1981).
Hu G, Heitmann JA, Rojas OJ. ‘Feedstock pretreatments strategies for producing ethanol from wood, bark, and forest residues’. BioResources 3, 2008, 270-94.
Saha BC, Cotta MA, ‘Enzymatic saccharification and fermentation of alkaline peroxide pretreated rice hulls to ethanol’. Enzyme Microbiol. Technol.,41, 2007,528-532.
Bhange V.P, Soni M.A., Kopulwar A.P. ‘Methane production from lignocellulosic biomass: a review’. International Jour. Of Emerging Trends in Engineering and Basic Sciences., 2349-6967, Vol.2, Issue 2, 2015, 68-74.
Jorgensen, H.; Kristensen, J. B.; Felby, C. ‘Enzymatic conversion of lignocellulose into fermentable sugars: Challenges and Opportunities’. Biofuels, Bioprod. Bioref., 1,2007,119–134.
Jorgensen, H.; Kristensen, J. B.; Felby, C. ‘Enzymatic conversion of lignocellulose into fermentable sugars: challenges and opportunities’. Biofuels, Bioprod. Bioref., 1,2007,119–134.
Sun, Y.,Cheng, J. ‘Hydrolysis of lignocellulosic materials for ethanol production: A review’. Bioresour. Technol., 83, 2002, 1–11.
Perez, J. Dorado, J. M. Rubia, T. D, Martinez, J. ‘Biodegradation and biological treatment of cellulose, hemicellulose and lignin: An overview’. Int. Microbiol. 5, 2002, 53–63.
McMillan, J. D. ‘Pretreatment of lignocellulosic biomass. in Enzymatic Conversion of Biomass for Fuels Production; Himmel, M. E., Baker, J. O., Overend, R. P. (Eds.; American Chemical Society: Washington, DC;1994)292-324.
Hsu, T.A., Ladisch, M.R., Tsao, G.T. ‘Alcohol from cellulose’. Chemical Technology, 10 (5), 1980,315–319.
De Sousa MV, Monteiro SN, d’Almeida JRM. ‘Evaluation of pretreatment size and molding pressure on flexural mechanical behavior of chopped bagasse–polyester composites’. Poly. Testing 23 (3), 2004, 253-258.
S. Xie, J.P. Frost, P.G. Lawlor, G. Wu, X. Zhan, ‘Effects of thermo-chemical pre-treatment of grass silage on methane production by anaerobic digestion’. Bioresource Technology, 102(19), 19, 2011, 8748-8755.
A. Lehtomäki, S. Huttunen, J.A. Rintala. ‘Laboratory investigations on co-digestion of energy crops and crop residues with cow manure for methane production: Effect of crop to manure ratio’. Resources, Conservation and Recycling, 51 (3),2007, 591-609.
K.S Jagadish, H.N Chanakya, P Rajabapaiah, V Anand. ‘Plug flow digestors for biogas generation from leaf biomass’. Biomass and Bioenergy,14,5–6,1998,415-421.
Liew Lo Niee, Shi Jian, Li Yebo. ‘Enhancing the solid-state anaerobic digestion of fallen leaves through simultaneous alkaline treatment’. Bioresource Technology,102(19),2011,8828-8834.
Lehtomaki, A., Huttunen, S., Lehtinen, T.M., & Rintala, J.A, ‘Anaerobic digestion of grass silage in batch leach bed processes for methane production’. Bioresource Technology, 99(8),2008,3267-3278.
Yu, H. W., Samani, Z., Hanson, A., & Smith, G. ‘Energy recovery from grass using two-phase anaerobic digestion’. Waste Management,22, 2002,1-5.
Parawira W, Read JS, Mattiasson B and Björnsson L. ‘Energy production from agricultural residues: high methane yields in pilotscale two-stage anaerobic digestion’. Biomass Bioenergy 32,2008,44–50.
Wilkie A, Goto M, Bordeaux FM and Smith PH. ‘Enhancement of anaerobic methanogenesis from napier grass by addition of micro-nutrients’. Biomass Bioenerg,11, 1986,135-146.
Jarvis A, Nordberg A, Jarlsvik T, Mathisen B and Svensson BH. ‘Improvement of a grass-clover silage-fed biogas process by the addition of cobalt’. Biomass Bioenerg,12,1997,453–460.
Nordberg A, Jarvis A, Stenberg B, Mathisen B and Svensson BH. ‘Anaerobic digestion of alfalfa silage with recirculation of process liquid’. Bioresource Technol 98, 2007, 104–111.