Top: Fluorine in solid form is primarily found in fluorite, a mineral. Fluorite rocks photo by Sally Brown
 Sally Brown
Sally Brown
BioCycle Editor Nora Goldstein recently forwarded me a press release about a new method for analyzing PFAS — Per- fluoroalkyl compounds —in soils and biosolids and asked if it was worth discussing in a column. The method was developed and released by ASTM (American Society for Testing and Materials). This is a reputable organization. Four biosolids were tested with four soils for PFAS. The combinations were tested at different relative concentrations. As PFAS analysis goes, this method claims to be relatively straightforward. The samples are extracted with a solvent, filtered, separated using liquid chromatography and then run through mass spectrometry. To see exactly which PFAS they have included and what the recoveries are you have to pony up $90 for the PDF.
This method is likely a step forward for those concerned about PFAS. It is not the final destination. PFAS analysis is highly complicated and confusing and constantly evolving. Four biosolids with four soils is like a drop in the ocean. It may be all the drop you need. That depends on how deep you want to dive.
A good way to understand what I mean is to talk about polyaromatic hydrocarbons (PAHs). Like PFAS, PAHs are a very broad group of compounds that have one thing in common. For PFAS it is carbon bonded to fluorine. For PAHs it is aromatic rings, or carbons bound to other carbons in a circular formation. Many PAHs are pretty innocuous. They degrade quickly and are no big deal. Examples that come to mind are compounds that you can find in wood smoke and when eating barbecued meats. The ones that you worry about are benzo A pyrene and the 16 or so others that are considered to be potentially toxic. There is a big difference between analyzing for all PAHs and analyzing for those that have been considered toxic. One has relevance from a toxicology perspective and the other can be a scare tactic.
Fingering The Bad Guys
With PFAS, we are not sure exactly what the bad guys are (granted we know that PFOA and PFOS fit into that category). As for the other 10,000+ or so varietals, the jury is still out. The other thing that makes this complicated is that recent research has shown that some of the not in theory bad guys can bond together to form evil doers during processes such as composting and anaerobic digestion.
Here are some examples from the literature to show what I mean. It is not possible to analyze for all of the types of PFAS that are out there. As a way to address this, authors have taken different approaches. In one widely cited paper on PFAS in food packaging, the authors analyzed for total fluorine rather than for specific compounds (Schaider et al., 2017). About 400 samples of paper that spent time around food were collected. That is a lot of fries, shakes and burgers. Fluorine was found in lots of them. Where it was present, it was generally found in (for these compounds) very high quantities. The authors tested for fluorine by using a type of spectroscopy, which gave them results in concentrations of fluorine per area of paper. (See “Fluorine And PFAS” at the end of the column to learn more about fluorine.)
A subset of the samples was also analyzed for specific PFAS compounds. Here the authors used a list of types of PFAS allowed for use in food packaging. In the supporting information for the paper, 25 of those compounds are listed. Now, if these authors had only tested for a certain subset of PFAS, results would likely have been different and potentially highly misleading. One could argue that by testing for the presence of fluorine rather than specific compounds, these results are misleading. As fluorine is something that you use in steel making, not for paper production (it is not a necessary plant nutrient and paper is typically made from wood fiber), I have no concerns about their approach, just about the food packaging.
What Is Forever?
Here is a second example. Linda Lee at Purdue University is to me (and many others) the best chemist working on PFAS in composts and biosolids. She recently presented work in process by one of her graduate students. Caroline Alukkal is looking at how the PFAS profile changes during anaerobic digestion. Most of the PFAS (according to Dr. Lee) coming into wastewater plants are short and are classified as diPaPs. PAP is short for Polyfluoroalkyl phosphate esters — try saying that 5 times fast. The ‘di’ refers to the fact that there are two esters. Esters are a class of organic compounds that react with water to form acids or alcohols. DiPAPs have been used in food packaging, personal care products, cleaning products, paints — basically all around your kitchens and bathrooms.
These di-PAPs are a category or class of PFAS compounds. There are a wide range of them. No idea if any or how many made the cut with the new ASTM testing protocol. The issue is what happens to all of these diPAPs during composting or anaerobic digestion. PFAS may be forever chemicals but diPAPs are not the form they like to take for all eternity. Different stabilization processes can transform these diPAPs into longer chain PFAS compounds, including the dreaded PFOS and PFOA. If you just analyze your wastewater influent for a few of the longer chain compounds, chances are you would find little to nothing (even with the parts per billion lens). Imagine the shock when you found higher quantities in your compost or digestate. In this example, analyzing for the full suite of diPAPs, intermediate and terminal products would allow you to understand the transformation process (Figure 1). It might be that shortening retention time in digesters allows you to limit this metamorphosis. Without knowing if the ASTM protocol takes this into account, it is hard to know if it gives you enough information to be useful.
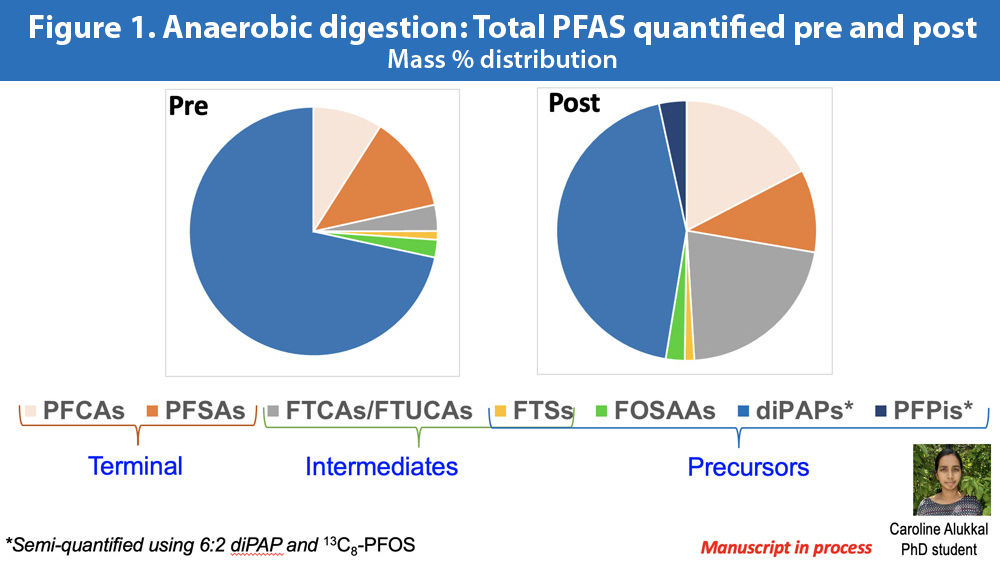 These are just two examples of potential concerns with this new protocol. Another to mention quickly is how well it will work across a wider range of biosolids, soils and analytical labs. Linda Lee mentioned that she had to get the cleaning staff where she works to change products. The PFAS in the cleaning products messed up her blanks and her recoveries. Andrew Carpenter at Northern Tilth, who has done much of the soil sampling for PFAS in Maine, said something similar. In his case it was the ceiling tiles in the lab that he was using that were contaminating his controls.
These are just two examples of potential concerns with this new protocol. Another to mention quickly is how well it will work across a wider range of biosolids, soils and analytical labs. Linda Lee mentioned that she had to get the cleaning staff where she works to change products. The PFAS in the cleaning products messed up her blanks and her recoveries. Andrew Carpenter at Northern Tilth, who has done much of the soil sampling for PFAS in Maine, said something similar. In his case it was the ceiling tiles in the lab that he was using that were contaminating his controls.
In a nutshell, when it comes to PFAS, we are talking about very, very small quantities of compounds and trying to get them teased out of materials that are complicated carbon mixtures. Unlike PAHs, we are not sure which of these we are really worried about. We also don’t fully understand that “forever” is a bit of a misnomer. These compounds change in different environments. The new method for PFAS analysis may be a step forward. It may be that over time, we will come to understand which types of PFAS we really care about. From my perspective, that is a long way off.
Sally Brown, BioCycle Senior Adviser, is a Research Professor at the University of Washington in the College of the Environment.



 In cases where carbon fluorine bonds are made, these bonds are very stable over a wide range of temperatures. They have been used as refrigerants but when the gas forms of these compounds are released to the atmosphere they have an enormous global warming potential, leaving methane in the distant dust. In other words, fluorine is not overly rare. In many forms it can be very helpful. It is primarily when we manufacture designer versions of it with different carbon bonds that we get into trouble.
In cases where carbon fluorine bonds are made, these bonds are very stable over a wide range of temperatures. They have been used as refrigerants but when the gas forms of these compounds are released to the atmosphere they have an enormous global warming potential, leaving methane in the distant dust. In other words, fluorine is not overly rare. In many forms it can be very helpful. It is primarily when we manufacture designer versions of it with different carbon bonds that we get into trouble.









